- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
20. July 2018
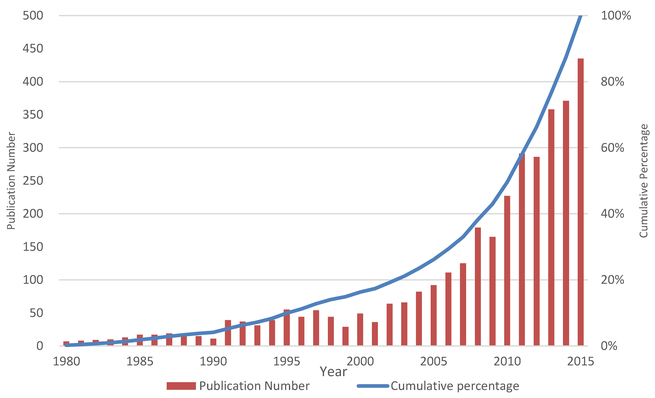
Solid dispersions are an effective formulation technique to improve the solubility, dissolution rate, and bioavailability of water-insoluble drugs for oral delivery. In the last 15 years, increased attention was focused on this technology. There were 23 marketed drugs prepared by solid dispersion techniques. Objective: This study aimed to report the big picture of solid dispersion research from 1980 to 2015. Method: Scientific knowledge mapping tools were used for the qualitative and the...
19. July 2018
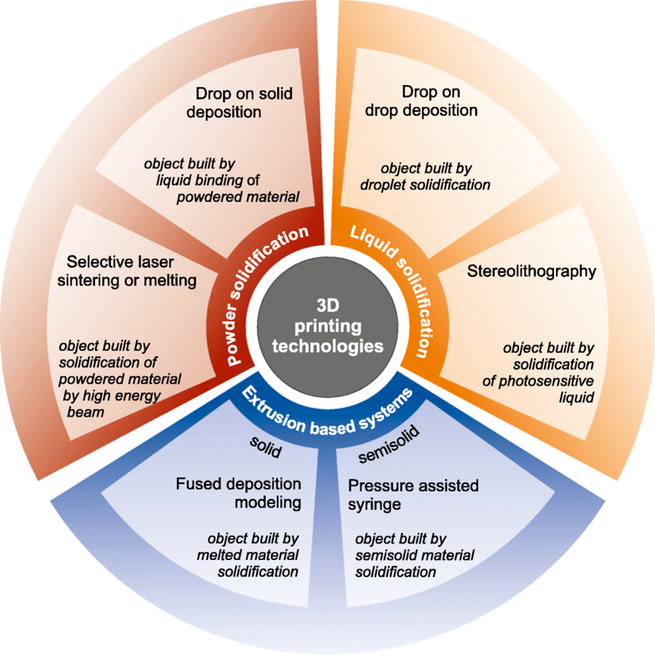
Growing demand for customized pharmaceutics and medical devices makes the impact of additive manufacturing increased rapidly in recent years. The 3D printing has become one of the most revolutionary and powerful tool serving as a technology of precise manufacturing of individually developed dosage forms, tissue engineering and disease modeling.
20. June 2018
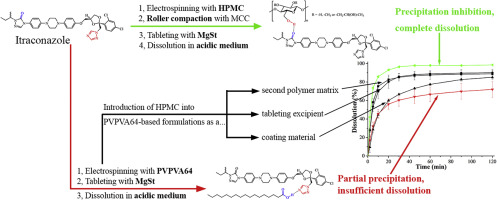
Itraconazole is a fungicide drug which has low bioavailability due to its poor water solubility. Amorphous solid dispersion (ASD) is a tool that has the potential to greatly increase the dissolution rate and extent of compounds. In this work, the dissolution of tablets containing the ASD of itraconazole with either hydroxypropyl methylcellulose (HPMC) or vinylpyrrolidone-vinyl acetate copolymer (PVPVA) was compared in order to find a formulation which can prevent the drug from the precipitation...
11. April 2018
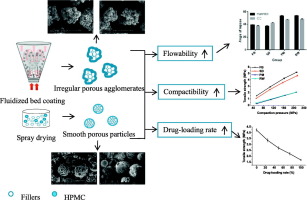
This study aimed to develop novel co-processed tablet fillers based on the principle of particle engineering for direct compaction and to compare the characteristics of co-processed products obtained by fluid-bed coating and co-spray drying, respectively. Water-soluble mannitol and water-insoluble calcium carbonate were selected as representative fillers for this study. Hydroxypropyl methylcellulose (HPMC), serving as a surface property modifier, was distributed on the surface of primary filler...
05. March 2018
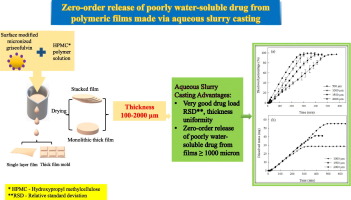
In spite of significant recent interest in polymeric films containing poorly water-soluble drugs, dissolution mechanism of thicker films has not been investigated. Consequently, release mechanisms of poorly water-soluble drugs from thicker hydroxypropyl methylcellulose (HPMC) films are investigated, including assessing thickness above which they exhibit zero-order drug release.
18. February 2018
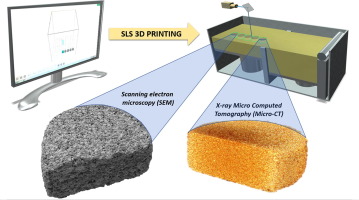
Abstract Selective laser sintering (SLS) is a three-dimensional printing (3DP) technology employed to manufacture plastic, metallic or ceramic objects. The aim of this study was to demonstrate the feasibility of SLS to fabricating novel solid dosage forms with accelerated drug release properties, and with a view to create orally disintegrating formulations. Two polymers (hydroxypropyl methylcellulose (HPMC E5) and vinylpyrrolidone-vinyl acetate copolymer (Kollidon® VA 64)) were separately...
15. February 2018
The poor oral bioavailability of many active pharmaceutical ingredients (APIs) resulting from low solubility is one of the important challenges in pharmaceutical technology. Over the last two decades the number of relatively insoluble drugs has grown steadily. Nowadays it is estimated that approximately 70% of new drug candidates are characterized by poor solubility.
07. February 2018
Polymer film blends of hydroxypropyl methylcellulose (HPMC) and amylose-sodium palmitate inclusion complexes (Na-Palm) were produced with no plasticizer, and were observed to have improved physical and gas barrier properties as compared with pure HPMC. The
30. January 2018
Many drugs are bitter and overcoming this bitter taste is a major barrier in developing a successful product, especially for pediatric patients. Approaches to mask taste include changing taste perception, creating a physical barrier to separate the drug from interacting with taste buds, and changing drug solubility.
29. January 2018
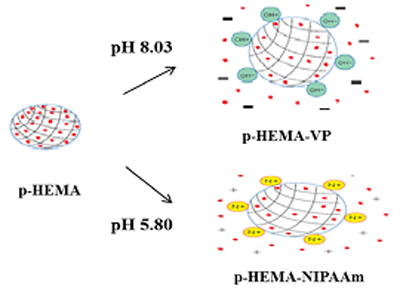
In this study, we evaluated contact lenses as drug delivery media and the effect of tear physiological pH on drug release behavior. Anionic or cationic hydroxyethyl methacrylate (HEMA) lenses were polymerized with either vinylpyrrolidone (VP) or N-isopropylacrylamide (NIPAAm). The pH of artificial tears was prepared as between 5.8 and 8.35. One of common treatments, hydroxypropyl methylcellulose (HPMC), to dry eye disease was used as a test drug.