- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
16. January 2018
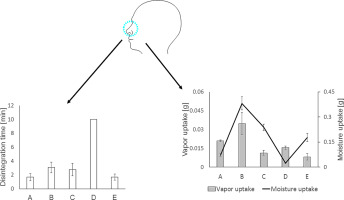
This present study intended to provide nasal adhesive formulations for the topical treatment of dry nasal syndrome. Mucoadhesive films were prepared according to solvent evaporation method consisting of well-known polymers such as gellan and carboxymethyl cellulose. Mucoadhesive films (A–E) were evaluated in respect to their physicochemical properties, stability, disintegration behavior and tensile strength.
27. December 2017
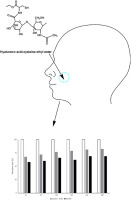
The aim of this study was to develop a solid dosage form for pediatric application. For this purpose, hyaluronic acid was covalently linked with sulfhydryl groups of cysteine ethyl ester via amide bond formation mediated by carbodiimide.
09. October 2017
A preclinical rabbit model of DED was created and animals were treated with the conceptually novel compound HASD to measure the clinical potential of this novel compound. While it performed well – at least as well as the best artificial tear solution Systane Ultra – this comparison is perhaps unfair to HASD
12. June 2017
Anionic polymers were modified with sulfhydryl bearing ligands to evaluate their mucoadhesiveness.
• Mucoadhesion augmented up to 59.23-fold compared to corresponding unmodified polymer.
• Tensile strength, bioadhesion, rotating cylinder and consolidation assay were performed on buccal tissue.