- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
21. September 2018
Glatt. Meet the Experts. CPHI 2018 in MADRID // Booth 4G41 Glatt Process Technology Pharma & Glatt Pharmaceutical Services We are already looking forward to welcoming you at CPHI Madrid // Booth 4G41 Award winning Glatt TwinPro® The first fusion of high-shear granulation and fluidized bed drying in the world
24. August 2018
A process control system based on PAT can compensate for variations in particle size, resulting in more consistent coating thickness.
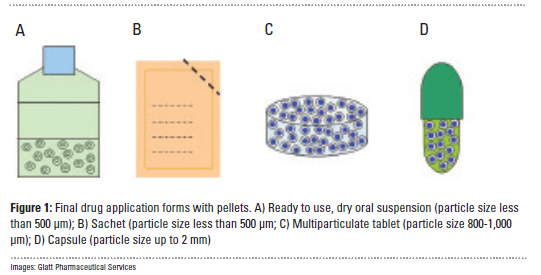
Drug-layered multiparticulates are a common dosage form for extended or modified-release pharmaceutical formulations. Delivered either in capsules, tablets, or as food additives in pediatric or geriatric applications (1), these formulations typically feature a functional coating designed to delay dissolution of the drug in the body.
18. August 2018
In contrast to classic single-unit dosage forms such as tablets, the dosage of the drug substance in multi-particulate systems is divided on a plurality of subunits – typically consisting of thousands of spherical pellet particles with a diameter of between 100 and 2,000 μm. This means that non-disintegrating, monolithic single-unit forms retain their structure in the digestive tract, whereas the multi-particular preparations consist of numerous sub-units which disperse after administration....
18. August 2018
Older adults with multimorbidity, polypharmacy, and complex health needs are the major consumer of health care. Ensuring that medicines are used safely, effectively, and delivered efficiently in this population is challenging. In this context, the approach to medicines delivery should seek to overcome some of the difficulties of delivering medicines to older people, and ensure each medication is delivered by the optimal and most convenient route for the patient in question. However, this poses...
13. August 2018
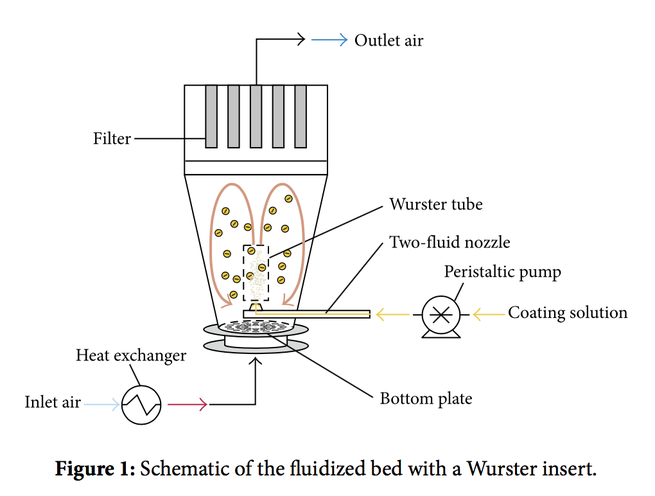
The aim of this study is to determine favorable process conditions for the coating of placebo tablets. Tablets made of microcrystalline cellulose are coated with hydroxypropyl cellulose polymer and Advantia™ Prime polymeric mixture film in lab-scale fluid-bed environment with a Wurster tube. In order to determine favorable process conditions (concentration, Wurster tube position, inlet air temperature, and atomization pressure), evaluation factors expressing process efficiency were...
13. July 2018
Neither a non-EU nor an EU company that have a VAT registration can easily use the VAT transit rule when importing drug products. This means that VAT has to be paid on imports of goods from outside the EU. Of course, this VAT can be reclaimed via the next monthly or quarterly VAT declaration. However, this means a cash flow disadvantage because import VAT needs to be pre-financed. Thus companies located in non-EU countries (e.g. Switzerland, USA, South America, Russia, etc.) planning to import...
17. April 2018
Corden Pharma in Plankstadt Germany trusts in Glatt Technology. In technological partnership with Weiss Pharmatechnik, Glatt provided state of the art pharma process solutions combined with top flexible barrier systems for contained developing service up to OEB 5 Level. Interested? Get in touch with Glatt
10. April 2018
"Glatt goes Compact and Modular" - Glatt Ingenieurtechnik has added compact continuous agglomeration equipment and new control systems to its "Modular Plant Engineering" Portfolio. At ACHEMA, the leading international trade fair for the process industries in June in Frankfurt am Main, market leading supplier of fluidized bed and spouted bed systems, Glatt, will showcase several new developments, including a compact system for continuous agglomeration and a modular control system that’s easy...
04. April 2018
A full day of lectures on the Basics of
Granulation, Wurster Coating, Tablet
Compression, Tooling and Troubleshooting
28. March 2018
Mini-tablets with diameters of 2.0, 2.5, and 3.0 mm are coated in two different lab-scale fluidized bed coaters equipped with a Wurster draft tube. The main focus of the research is to evaluate the inter-particle coating variability, and to assess the contribution of cycle time variation. Cycle times are measured using a photoluminescent tracer with a detector mounted on the top of the draft tube. The number of passes variability is represented from 5 to 28% of the total coating variability....