- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
24. August 2018
A process control system based on PAT can compensate for variations in particle size, resulting in more consistent coating thickness.
Drug-layered multiparticulates are a common dosage form for extended or modified-release pharmaceutical formulations. Delivered either in capsules, tablets, or as food additives in pediatric or geriatric applications (1), these formulations typically feature a functional coating designed to delay dissolution of the drug in the body.
26. June 2018
More and more pharmaceutical products are reaching the market as multiparticulate dosage forms, mainly as pellets. The healthcare sector also frequently selects pellets as the optimal marketable form of functional food. As there are quite a number of existing techniques relating to the production of pellets, it is often very difficult for a formulator or marketing manager to make a choice, since every technique claims to be optimal.
22. November 2017
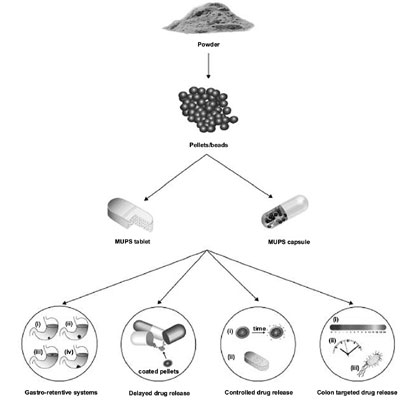
Background and Objective: Single-unit solid oral dosage forms such as tablets and capsules are considered the most common and acceptable form of immediate release systemic drug delivery systems. On the other hand, multiple-unit pellet systems (MUPS) have in recent years become an important dosage form that offers various advantages over conventional single-unit solid oral dosage forms.
23. August 2017
The multiparticulate drug product concept covering micropellets, pellets, and mini-tablets is presented as a highly feasible approach to present convenient and patient friendly medication for the geriatric population. Improved swallowability and optimized administration regimen going along with defined drug dosage are achievable.