- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
13. September 2018
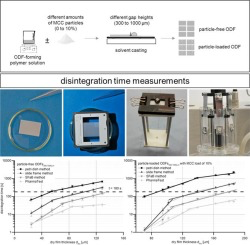
Orodispersible films (ODFs) provide high application comfort due to rapid disintegration in the oral cavity. They increasingly found the approval of pharmaceutical research and development and were added to the European Pharmacopeia in 2012. The European Pharmacopeia explicitly demands disintegration testing for ODFs, but does not refer to a suitable method. The aim of this study was to collect and evaluate existing disintegration methods regarding their suitability to investigate different ODF...
13. September 2018
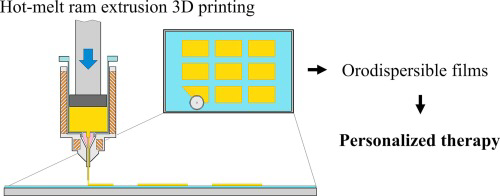
This work demonstrates the feasibility of the extemporaneous preparation of maltodextrins orodispersible films (ODF) by hot-melt ram-extrusion 3D printing. This method consists of three simple technological operations which can be also implemented in a pharmacy setting. First, maltodextrins, drug, and other excipients are mixed in a mortar and wetted with the plasticizer (i.e. glycerine). Then, the mixture is fed in the chamber of the ram-extruder and heated. ODF are individually printed on the...
07. June 2018
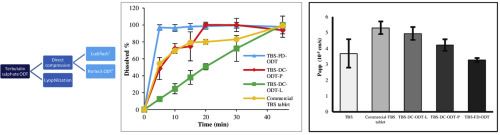
Asthma is a chronic respiratory condition characterized by attacks of spasm in the bronchi of the lungs, causing difficulty in breathing. Oral and inhalation routes are generally used for the treatment of asthma. Terbutaline sulfate (TBS), is a widely used bronchodilator for the treatment of asthma, is available in formulations in the market. However, there is no commercially available orally disintegrating tablets (ODTs) containing TBS. Therefore, this study was aimed to develop and...
28. May 2018
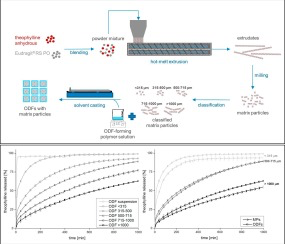
Orodispersible films (ODFs) are an advantageous dosage form to accomplish patient convenience and compliance in oral drug delivery. They provide a number of special application features, such as the ease of administration without water and suitability for patients with swallowing problems. However, this promising dosage form has been limited to immediate release formulations so far. The aim of this study was to develop a thin film produced by solvent casting, which is rapidly disintegrating...
08. May 2018
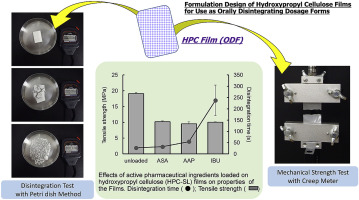
Hydroxypropyl cellulose (HPC) is a water-soluble polymer used as a binder during pharmaceutical tableting and granulation. HPC is also known as a base material for pharmaceutical film by virtue of its film formability with excellent plasticity. The aim of this study was to assess the applicability of HPC to orally disintegrating film (ODF) and to investigate optimization of the ODF formulation of HPC. The effects of the molecular weight of HPC and the addition of active pharmaceutical...
21. October 2017
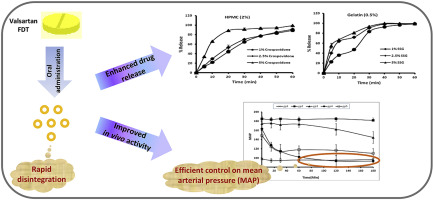
Fast disintegrating tablets (FDTs) dissolve or disintegrate in the mouth without the need of additional water. The aim of the present study was to formulate FDTs of Valsartan for the treatment of hypertension in children who could find difficulties in swallowing conventional solid dosage forms. The tablets were prepared by wet granulation technique. Superdisintegrants such as sodium starch glycolate (SSG) and crospovidone were optimized as 5% on the basis of least disintegration time. Different...
03. September 2017
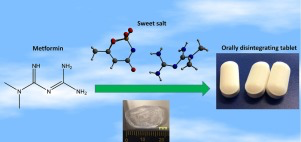
Abstract Salt formation has been intensively used to improve drug properties, including solubility, stability and mechanical properties. A sweet salt of metformin with acesulfame, prepared though an anion exchange reaction, showed superior properties over the commercial hydrochloride salt. These included both remarkable improvement of taste and significant enhancement in tabletability, which is explained by the different crystal structures and lower hardness as measured by nanoindentation. The...
22. April 2017
Abstract Mini-tablets have potential applications as a flexible drug delivery tool in addition to their generally perceived use as multi-particulates. That is, mini-tablets could provide flexibility in dose finding studies and/or allow for combination therapies in the clinic. Moreover, mini-tablets with well controlled quality attributes could be a prudent choice for administering solid dosage forms as a single unit or composite of multiple mini-tablets in patient populations with swallowing...
20. April 2017
Abstract Optimized orally disintegrating tablets (ODTs) containing furosemide (FUR) were prepared by direct compression method. Two factors, three levels (32) full factorial design was used to optimize the effect of taste masking agent (Eudragit E100; X1) and superdisintegarant; croscarmellose sodium (CCS; X2) on tablet properties. A composite was prepared by mixing ethanolic solution of FUR and Eudragit E100 with mannitol prior to mixing with other tablet ingredients. The prepared ODTs were...
20. April 2017
Abstract Objective: The aim of this work was to investigate the potential of controlled precipitation of flurbiprofen on solid surface, in presence or absence of hydrophilic polymers, as a tool for enhnaced dissolution rate of the drug. The work was extended to develop rapidly disintegrated tablets. Significance: This strategy provide simple technique for dissolution enhancement of slowly dissolving drugs with high scaling up potential. Method: Aerosil was dispersed in ethanolic solution of...