- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
20. September 2018
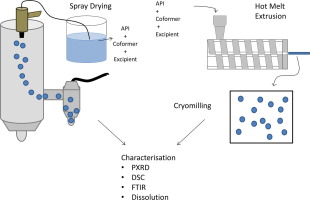
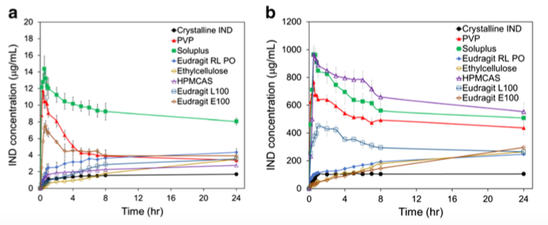
The comparison of spray drying versus hot melt extrusion (HME) in order to formulate amorphous solid dispersions has been widely studied. However, to the best of our knowledge, the use of both techniques to form cocrystals within a carrier excipient has not previously been compared. The combination of ibuprofen (IBU) and isonicotinamide (INA) in a 1:1 molar ratio was used as a model cocrystal. A range of pharmaceutical excipients was selected for processing - mannitol, xylitol, Soluplus and PVP...
09. March 2018
The use of amorphous solid dispersions (ASD) to overcome poor drug solubility has gained interest in the pharmaceutical industry over the past decade. ASDs are challenging to formulate because they are thermodynamically unstable, and the dispersed drugs tend to recrystallize. Until now, most research on ASDs has focused on immediate-release formulations, supersaturation, and stability; only a few studies have recently reported on the manufacturing of sustained-release ASDs.
27. February 2018
Novel medicated straws were developed based on drug-loaded
electrospun fibers prepared by direct current electrospinning
(DCES) and high-speed electrospinning (HSES) of scaled-up
productivity. Good quality micro- and nanofibers were
electrospun using both techniques despite the multiple times
higher throughput rate of HSES based on the scanning electron
microscopic imaging (SEM). Solid state analyses revealed that
the poorly soluble model drug carvedilol (CAR) was dispersed
in an amorphous form
11. January 2018
◾Amorphous solid dispersions
◾Amorphous-amorphous phase separation
◾Solvent selection for ASD preparation
◾Thermodynamic phase behaviour
22. May 2017
Nitrendipine amorphous solid dispersions named TDP-PVP and TDP-PEG were prepared.
Hydrogen bond forces were formed between drug and excipient in TDP-PVP and TDP-PEG.
Fit profile of contact angle to get dissolution mechanism of TDP-PVP and TDP-PEG.
TDP-PEG was carrier-controlled and TDP-PVP was drug/carrier-controlled diffusion