- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
05. March 2018
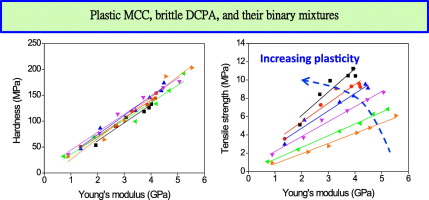
Mechanical properties of pharmaceutical materials, e.g., Young's modulus (E), indentation hardness (H), and tensile strength (σ), play an important role in powder compaction process. However, few studies investigated the relationship among these parameters and consequence for tablet compression. Using microcrystalline cellulose, a plastic material, and dibasic calcium phosphate anhydrate, a brittle material, as well as their binary mixtures, we systematically examined the relationship among the
16. January 2018
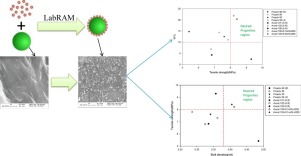
We adopt a Quality by Design (QbD) paradigm to better control the mechanical prop- erties of tablets. To this end, the effect of particle size distribution, lubricant concentra- tion, and mixing time on the tensile strength and elastic modulus of tablets is studied. Two grades of lactose, monohydrate and spray-dried, are selected. Tablets are compressed to different relative densities ranging from 0.8 to 0.94 using an instrumented compaction simulator.
03. December 2017
Excipients with good flowability, bulk density as well as compaction properties are desired for use in tableting since they play important roles in formulation development and processing, including, handling, mixing, feeding and compaction.
17. October 2017
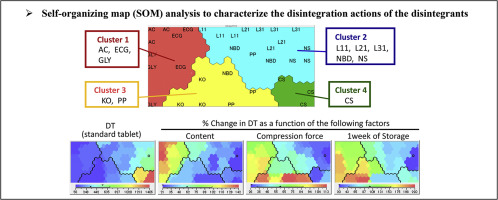
To gain a better understanding of disintegration actions, 11 different disintegrants were tested. Model tablets were prepared with various preparation conditions, and their disintegration time and tensile strength were measured.