- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
30. July 2018
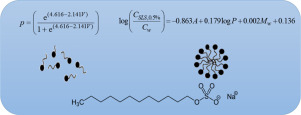
Micellar solubilization is a great method for increasing drugs solubility in aqueous environments. At concentrations above the critical micelle concentration (CMC), micelles are formed and they are able to increase the apparent aqueous solubility of poorly soluble drugs. Sodium lauryl sulfate (SLS) is one of the common solubilizing agents in pharmaceutical sciences. Investigation on the water solubility of drugs in the presence of surfactants and the development of a relationship between drug...
27. July 2018
The present study elucidated the advantages of using hydroxypropyl methylcellulose E5 (HPMC) as auxiliary excipient in maintaining storage stability and solubilization ability for curcumin amorphous solid dispersion (Cur ASDs) formulated by Eudragit E100 (E100). Polarized light microscopy and in vitro dissolution experiment was applied for confirming the ability of HPMC on inhibiting crystallization thereby maintaining storage stability in Cur ASDs. Meanwhile, as a non-ionic surfactant, HPMC...
18. June 2018
Gelucire® is Gattefossé’s brand of semi-solid excipients originally designed for hard gelatin capsule molding. The name results from the fusion between « gélule » meaning capsule and « cire » meaning wax. Following the name, there is a set of two figures: the first (eg 44) indicates the melting point in Celsius degree, the second (eg 14) indicates the theoretical HLB (Hydrophilic Lipophilic balance). The family is composed of four products with their own characteristics, specificities...
19. April 2018
Cell-free permeation systems are gaining interest in drug discovery and development as tools to obtain a reliable prediction of passive intestinal absorption without the disadvantages associated with cell- or tissue-based permeability profiling. Depending on the composition of the barrier, cell-free permeation systems are classified into two classes including (i) biomimetic barriers which are constructed from (phospho)lipids and (ii) non-biomimetic barriers containing dialysis membranes. This...
03. November 2017
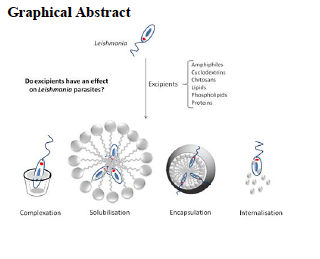
Leishmaniasis is a neglected tropical disease responsible for the ninth largest disease burden in the world. Excipients are necessary for ensuring the stability and bioavailability of currently available antileishmaniasis drugs.
10. October 2017
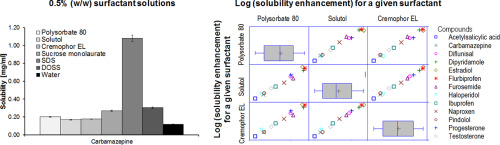
Solubility screening in different surfactant solutions is an important part of pharmaceutical profiling. A particular interest is in low surfactant concentrations that mimic the dilution of an oral dosage form.
01. September 2017
The aim of the present work was to prepare tadalafil (TDF) nanocrystals-loaded oral polymeric films (OFs) and investigate the effect of hydrophilic surfactants and drug loads on the physico-mechanical and dissolution properties.
24. July 2017
Tadalafil suffers from poor aqueous solubility that could lead to fluctuating blood levels and unreproducible effect. Thus, this work aimed at improving tadalafil dissolution utilizing the approach of surfactant-enriched tablets.
22. June 2017
Discovery of several poorly water soluble drugs in the past decade has led to the need of developing a novel dosage form which increases the solubility of the drug and improves oral bioavailability.
22. November 2016
Abstract Gastrointestinal drug administration is the preferred route for the majority of drugs however, the natural physiology and physicochemistry of the gastrointestinal tract is critical to absorption but complex and influenced by factors such as diet or disease. The pharmaceutical sciences drive for product consistency has led to the development of in vitro product performance tests whose utility and interpretation is hindered by the complexity, variability and a lack of understanding. This...