- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
04. April 2018
During the development of parenteral dosage forms, different physicochemical studies are required to ensure stable, effective and safe formulations. The osmolality of this kind of dosage form should bear a close similarity to the body fluids to prevent local irritation, pain or even more significant side effects like endothelial damage. The osmotic studies performed in Polyethylene glycol 400 (PEG 400), Polyethylene glycol 4000 (PEG 4000), Poloxamer 407 (P407), Sodium Hyaluronate (SH),...
15. February 2018
The poor oral bioavailability of many active pharmaceutical ingredients (APIs) resulting from low solubility is one of the important challenges in pharmaceutical technology. Over the last two decades the number of relatively insoluble drugs has grown steadily. Nowadays it is estimated that approximately 70% of new drug candidates are characterized by poor solubility.
14. February 2018
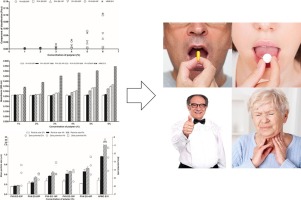
Solid oral dosage forms (SODF) are drug vehicles commonly prescribed by physicists in primary and secondary cares, as they are the most convenient for the patient and facilitate therapy management. Concerns regarding unintended adhesion of SODF during oro-esophageal transit remain, especially in multimorbid patients, bedridden patients and patients suffering from dysphagia.
09. February 2018
Preparation of mini buccal tablets incorporating either of the Alzheimer’s drugs, rivastigmine tartrate or donepezil hydrochloride, were developed for patients who have difficulty swallowing as a source of effervescence, by pairing the less commonly used malic acid with sodium bicarbonate in an experimentally determined (1:2) stochiometirc ratio.
23. January 2018
Glucagon is a peptide hormone used for the treatment of hypoglycemia; however, its clinical potential is limited by its insolubility and instability in solution. Herein, the encapsulation, stabilization, and release of glucagon by trehalose glycopolymer nanogels are reported. Methacrylate-functionalized trehalose is copolymerized with pyridyl disulfide ethyl methacrylate using free radical polymerization conditions to form trehalose glycopolymers with thiol-reactive handles.
10. January 2018
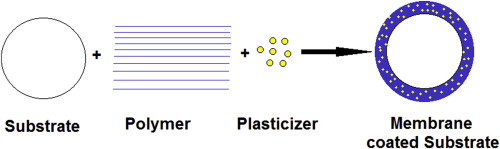
Extended drug delivery offers numerous benefits compared to immediate release dosage form. Membrane coating of immediate release dosage form is an effective means of producing an extended drug delivery system. Polymers when coated alone, have limited ability to form the rate controlling membrane with good mechanical properties.
13. December 2017
Polyethylene glycol (PEG) is a chemically inert, amphiphilic polymer used as an excipient in many pharmaceuticals for decades, and as a conjugate with biologicals (ie, PEGylation) since 1990. To date, the US FDA has approved 14 PEGylated therapies with diverse indications.
09. December 2017
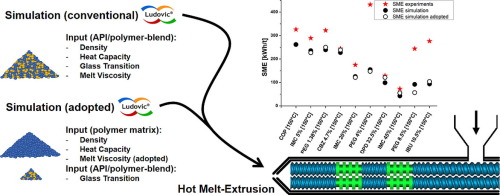
Simulation of HME processes is a valuable tool for increased process understanding and ease of scale-up. However, the experimental determination of all required input parameters is tedious, namely the melt rheology of the amorphous solid dispersion (ASD) in question.
13. November 2017
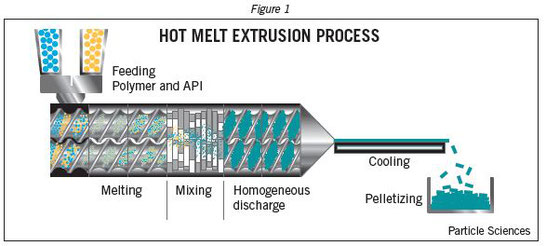
Amorphous solid dispersions (SDs) are considered as one of the most effective strategies for the formulation of poorly water-soluble compounds. The active compound is dispersed in an inert carrier composed of a polymer and active excipients. Since the drug is amorphous, there is typically an increase in apparent solubility as well as dissolution rate. Various methods are employed for manufacturing of SDs, nevertheless, hot-melt extrusion (HME) has become one of the most common process techniques
31. October 2017
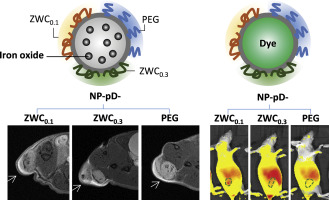
For polymeric nanoparticles (NPs) to deliver more drugs to tumors than free drug solution, it is critical that the NPs establish interactions with tumor cells and avoid removal from the tumors. Since traditional polyethylene glycol (PEG) surface layer interferes with the cell-NP interaction in tumors, we used a water-soluble and blood-compatible chitosan derivative called zwitterionic chitosan (ZWC) as an alternative surface coating for poly(lactic-co-glycolic acid) (PLGA) NPs.