- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
10. April 2018
The pharmaceutical industry has historically taken a cautious approach to new advancements. But now, it is embracing the possibilities of continuous manufacturing technologies. While traditional batch based processes are still the industry standard, there are several products on the market that have utilized continuous manufacturing practices in their development. Over the last decade, this approach has generated a considerable amount of attention. To take a deeper look at the issues...
05. March 2018
This study presents a framework for process and product development on a continuous direct compression manufacturing platform. A challenging sustained release formulation with high content of a poorly flowing low density drug was selected. Two HPMC grades were evaluated as matrix former: standard Methocel CR and directly compressible Methocel DC2. The feeding behavior of each formulation component was investigated by deriving feed factor profiles.
08. February 2018
In the highly regulated pharmaceutical industry, manufacturers can’t help but wonder what regulatory authorities like the FDA think of emerging technologies. So what do the global authorities have to say about continuous manufacturing?
12. October 2017
The paradigm shift in the pharmaceutical industry to continuous manufacturing, which has recently progressed from conceptual demonstration to pilot production, has stimulated the development and application of process systems engineering (PSE) tools for implementing efficient and robust control strategies.
03. July 2017
Industry delegates say the US Food and Drug Administration (FDA) is showing increased support for continuous manufacturing, a stance C-SOPS told us is quality control-driven.
29. June 2017
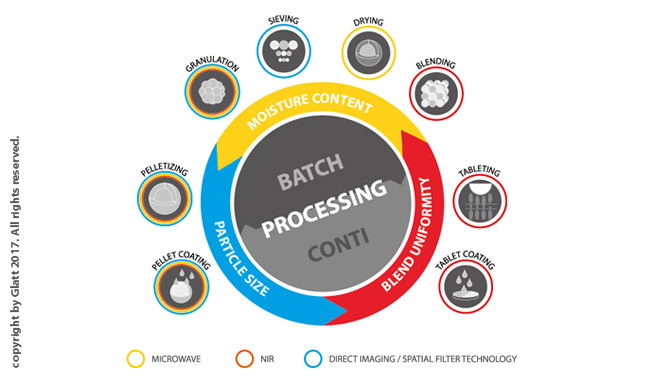
Process Analytical Technology, or "PAT" is the term given to analytical instruments developed to measure certain attributes of product within the manufacturing process, eliminating, or substantially minimising the need for sampling for off-line analysis
19. June 2017
16 hours hands-on seminar on continuous OSD pharma processing
02. May 2017
Live Presentations of MODCOS Direct Compression @ Interpack:
May 4/5/8 /9: 12.00 @ Glatt -Booth Hall 16 B03
24. August 2016
Abstract The present study summarizes the experimental characterization of a new continuous powder mixer (GCG-70 by Glatt®) using common pharmaceutical ingredients. The powder hold-up and residence time distribution were used to characterize the bulk behavior of the mixer as a function of impeller rotational speed, total throughput (mass flow rate) and blade configuration. The relative standard deviation (RSD), calculated from samples taken at the outlet of the blender, was used to...
23. September 2015
With the Glatt MODCOS (Modular Continuous System) process insert, batch versions of the GPCG 2 and GPCG 10 series can be easily converted to continuous fluid bed systems and integrated into a continuous process chain. More Information & Video