- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
22. March 2018
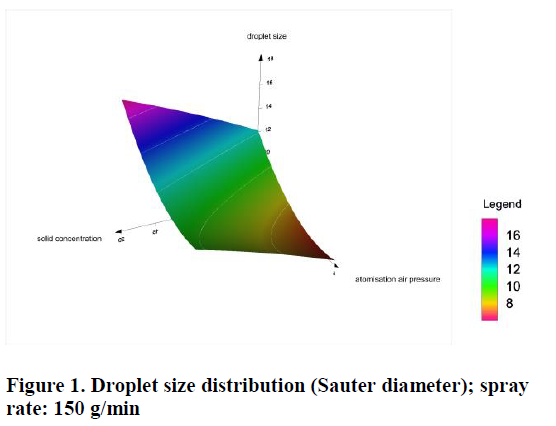
To obtain a reproducible and specification conforming product quality it is mandatory to control the process parameters for granulation and layering or coating. One important parameter for the development of coating and granulation processes in high shear and fluid bed processes is the spraying system [1] e. g. the application of a feasible droplet size coming out of the respective spraying nozzle. Too large droplets could result in oversized granules or in pellets´ agglomeration, whereas too...
19. January 2018
The authors studied six different grades of HPC to determine how they performed in high-shear and fluid-bed granulation.
18. November 2017
by Innopharmatechnology & Glatt @ PMEC India 2017 Learn about the latest Process Analytical Technology (PAT) technical and application developments for in-line PSD and Moisture measurement in fluid bed batch and continuous powder processes. More
21. September 2017
The objective of this study was to prepare time-controlled release etodolac pellets to facilitate drug administration according to the body’s biological rhythm, optimize
the drug’s desired effects, and minimize adverse effects.
19. September 2017
Fluid bed coating offers potential advantages as a formulation platform for amorphous solid dispersions (ASDs) of poorly soluble drugs, being a one-step manufacturing process which could reduce therisk of phase separation associated with multiple step manufacturing approaches. However, the impact of the physicochemical nature of carrier spheres on the properties and drug release from the ASDshas not been studied in detail. In this work, tartaric acid (TAP) and microcrystalline cellulose (CEL)...
11. September 2017
The purpose of this work was to develop multi unit particulate formulation using fluid bed processor for the controlled release of Metoprolol succinate and to understand the impact of formulation parameters on the critical quality attributes using a quality-by-design approach.
23. August 2017
The multiparticulate drug product concept covering micropellets, pellets, and mini-tablets is presented as a highly feasible approach to present convenient and patient friendly medication for the geriatric population. Improved swallowability and optimized administration regimen going along with defined drug dosage are achievable.
10. July 2017
Poster by Glatt Pharmaceutical Services & Credentis AG @EUPFI Congress in Warshaw -
22. February 2016
MARCH 6 - 9, 2016 - Long Island University, Brooklyn, NY LECTURES & LABORATORY EXERCISES WILL COVER Formulation Development • Modified Release Technologies Tablet Tooling Design • Tablet Scale Up Issues with HANDS-ON LEARNING where YOU OPERATE the equipment!
07. January 2016
Compaction of multiple-unit pellet system (MUPS) tablets has been extensively studied in the past few decades but with marginal success. This study aims to investigate the formulation and process strategies for minimizing pellet coat damage caused by compaction and elucidate the mechanism of damage sustained during the preparation of MUPS tablets in a rotary tablet press. More