- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
19. June 2018
Symposion with First-hand knowledge related to DPI technology along the complete process chain (from powder characteristics and formulations, filling technologies, device design assembly and packaging)
Discussions on the latest market trends and “state of the art” insights with regard to quality control, excipients and manufacturing Technology.
23. April 2018
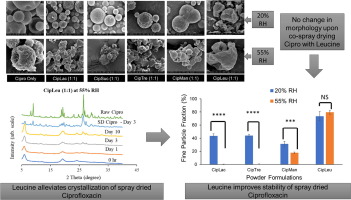
The aim of this study is to investigate the influence of excipients on physical and aerosolization stability of spray dried Ciprofloxacin dry powder inhaler formulations. The model drug, Ciprofloxacin hydrochloride, was co-spray dried with excipients such as disaccharides (sucrose, lactose, trehalose), mannitol and L-leucine. The spray dried samples were stored at two different relative humidity (RH) conditions of: (1) 20% and (2) 55% RH at 20°C. Ciprofloxacin co-spray dried with disaccharides...
01. April 2018
Amorphous powders are thermodynamically unstable, significantly impacting the processing, storage and performance of a product. Therefore, stabilization of the amorphous contents is in demand.
06. March 2018
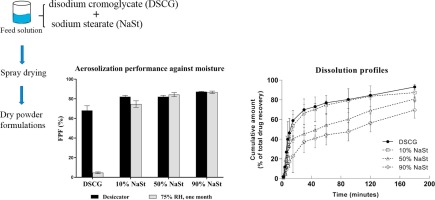
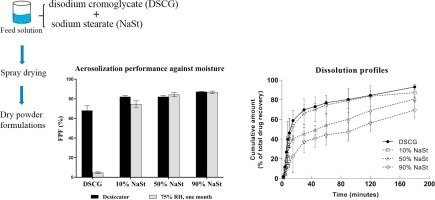
Amorphous powders are thermodynamically unstable, significantly impacting the processing, storage and performance of a product. Therefore, stabilization of the amorphous contents is in demand. In this study, disodium cromoglycate (DSCG) powder was chosen as a model drug because it is amorphous and highly hygroscopic after spray drying.
20. December 2017
The potential of the force control agent (FCA) magnesium stearate (MgSt) to enhance the aerosol performance of lactose-based dry powder inhaled (DPI) formulations was investigated in this study. The excipient blends were investigated with analytical techniques including time-of-flight secondary ion mass spectrometry (ToF-SIMS) and Single Particle Aerosol Mass Spectrometry (SPAMS) and particle size, morphology and surface properties were evaluated.