- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
29. August 2018
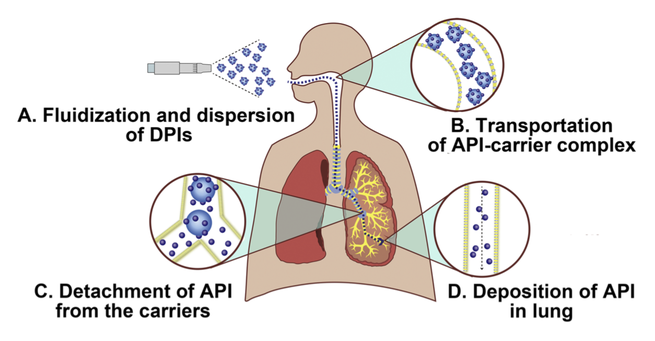
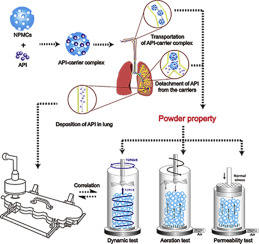
Carrier based dry powder inhalers (DPIs) are common vehicles for pulmonary delivery of active pharmaceutical ingredients (APIs), thus powder properties of carrier would generate a pronounced impact on the two dominant delivery stages of the DPIs. In our previous study, a novel modified carrier, the nanoporous mannitol carriers (NPMCs) was prepared to achieve satisfactory aerosolization performance. While the aerosolization performance enhancement mechanism of NPMCs based DPI was still...
28. August 2018
Carrier based dry powder inhalers (DPIs) are common vehicles for pulmonary delivery of active pharmaceutical ingredients (APIs), thus powder properties of carrier would generate a pronounced impact on the two dominant delivery stages of the DPIs. In our previous study, a novel modified carrier, the nanoporous mannitol carriers (NPMCs) was prepared to achieve satisfactory aerosolization performance. While the aerosolization performance enhancement mechanism of NPMCs based DPI was still...
19. June 2018
Symposion with First-hand knowledge related to DPI technology along the complete process chain (from powder characteristics and formulations, filling technologies, device design assembly and packaging)
Discussions on the latest market trends and “state of the art” insights with regard to quality control, excipients and manufacturing Technology.
08. February 2018
Development of bi-component generic orally inhaled product (OIP) delivered from dry powder inhaler (DPI) is challenging due to the necessity to demonstrate the similarity of size distribution of both APIs in inhaled aerosol. The effects of selected technical factors on OIP development are investigated. The Monodose inhalers with different aerodynamic resistance were used to aerosolize generic fluticasone/salmeterol formulations. Flow dynamics in the DPIs was analyzed during realistic use. The...
01. February 2018
Inhalation therapy is popular to treat lower respiratory tract infections. Azithromycin is effective against some bacteria that cause respiratory tract infections; but it has poor water solubility that may limit its efficacy when administrated as inhalation therapy. In this study, dry powder inhaler formulations were developed by co-spray drying azithromycin with L-leucine with a purpose to improve dissolution.
17. October 2017
Dry powder inhaler for pulmonary drug delivery: human respiratory system, approved products and therapeutic equivalence guideline
16. October 2017
More and more therapeutic proteins are developed for an administration by inhalation to treat respiratory diseases. PEGylation is an interesting approach for sustaining the residence time of these biopharmaceuticals in the lungs and thereby decrease the frequency of administration and the daily burden of inhalation therapies.