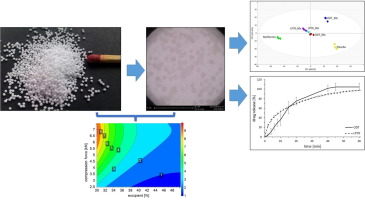
Compaction of multiparticulates into tablets, particularly into orodispersible tablets (ODTs), is challenging. The compression of pellets, made by solid lipid extrusion/spheronization processes, presents peculiar difficulties since solid lipids usually soften or melt at relatively low temperature ranges and due to applied mechanical forces. Until now, there are no reports in literature about the development of ODTs based on solid lipid pellets. To investigate the feasibility of producing such tablets, a design of experiment (DoE) approach was performed to elucidate the influence of compression force and amount of two co-processed excipients (Ludiflash® and Parteck® ODT) on properties of the tablets (friability, tensile strength, and disintegration time). ODTs (15 mm, flat-faced) with solid lipid pellets (250-1000 µm in diameter) containing 500 mg of metformin HCl, presenting immediate drug release profile and taste-masked properties, were targeted. During compression, a strong lamination of the tablets containing Parteck® ODT was observed. This phenomenon was prominently observed when high compression forces (≥ 5 kN) and high excipient amounts (≥ 40%; w/w) were used. On the other hand, the DoE focused on tablets with Ludiflash® showed better results regarding the production of ODTs. A positive influence of the compression force on the tensile strength, friability, and disintegration time of the tablets, regarding specifications of the Ph. Eur., was observed. The increase in the amount of this excipient resulted in fast disintegrating tablets, however, a negative influence on the tensile strength was noticed. After optimization of the parameters and formulation, based on the DoE results and considering the Ph. Eur. specifications for tablets, ODTs based on lipid pellets containing metformin HCl presenting immediate release profile (85% drug release in less than 30 min) and taste-masked properties (determined by an electronic tongue) were successfully obtained.
- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact