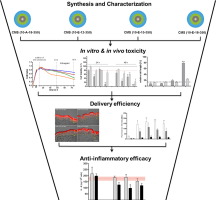
Drug loaded dendritic core-multishell (CMS) nanocarriers are of especial interest for the treatment of skin diseases, owing to their striking dermal delivery efficiencies following topical applications. CMS nanocarriers are composed of a polyglycerol core, connected by amide-bonds to an inner alkyl shell and an outer methoxy poly(ethylene glycol) shell. Since topically applied nanocarriers are subjected to biodegradation, the application of conventional amide-based CMS nanocarriers (10-A-18-350) has been limited by the potential production of toxic polyglycerol amines. To circumvent this issue, three tailored ester-based CMS nanocarriers (10-E-12-350, 10-E-15-350, 10-E-18-350) of varying inner alkyl chain length were synthesized and comprehensively characterized in terms of particle size, drug loading, biodegradation and dermal drug delivery efficiency. Dexamethasone (DXM), a potent drug widely used for the treatment of inflammatory skin diseases, was chosen as a therapeutically relevant test compound for the present study. Ester- and amide-based CMS nanocarriers delivered DXM more efficiently into human skin than a commercially available DXM cream. Subsequent in vitro and in vivo toxicity studies identified CMS (10-E-15-350) as the most biocompatible carrier system. The anti-inflammatory potency of DXM-loaded CMS (10-E-15-350) nanocarriers was assessed in TNFα supplemented skin models, where a significant reduction of the pro-inflammatory cytokine IL-8 was seen, with markedly greater efficacy than commercial DXM cream.
In summary, we report the rational design and characterization of tailored, biodegradable, ester-based CMS nanocarriers, and their subsequent stepwise screening for biocompatibility, dermal delivery efficiency and therapeutic efficacy in a top-down approach yielding the best carrier system for topical applications.